EFFECTS OF SHEWANELLA PUTREFACIENS PDP11 ON SENEGALESE SOLE SKELETOGENESIS
Introduction
Solea senegalensis is a highly valuable commercial fish. Skeletal abnormalities are a serious economical problem in sole aquaculture that may suppose about 15-40% of production discards. Nutrition during early life stages plays a key role in the morphogenesis and skeletogeneis of the larvae (Boglione et al., 2013). In this sense probiotic supplementation has been reported to improve skeletal development in rainbow trout and seabass larviculture (Aubin et al., 2005; Lamari et al., 2013). Shewanella putrefaciens Pdp11 bioencapsulated in Artemia (10-86 dah) enhances sole production parameters (Lobo et al., 2014). The aim of this study was to evaluate the effect of two different S. putrefaciens Pdp11 probiotic pulses using Artemia and rotifer (2-21 dah) or only Artemia (10-21 dah) as live vectors on the presence and severity of skeletal abnormalities in post-weaned (66 dah) Senegalese sole juveniles.
Methods
S. senegalensis larvae (40 L-1) were distributed into 280 l circular polyester rearing tanks. Temperature was 18.1 ± 1.1 ºC along the trial. Illumination (1000 lux on surface water) was continuous till 10 dah and a 0:24 L:D cycle (< 20 lux) was established from 17 dah onwards. Feeding regime was based on Cañavate and Fernández-Díaz (1999) with modifications. Phytoplankton and rotifers were supplied (2-9 dah) and cofeeding with Artemia metanauplii (Origreen, Skretting) and dry feed (Start-Larviva, Biomar) was made after 10 dah. Weaning (Wean Ex-Larviva, Biomar) started at 48 dah and finished at 66 dah. S. putrefaciens Pdp11 was daily incubated in TSA (1.5%NaCl) at 22ºC, collected and suspended in a PBS solution (pH 7.2) and finally supplied to rotifers or Artemia (2.5 107 cfu mL-1) three hours prior to larval feeding. Three experimental groups were established: a) CCC Control, b) CPC fed with S. putrefaciens Pdp11 supplemented Artemia (10-21 dah) and c) PPC S. putrefaciens Pdp11 supplemented rotifer and Artemia (2-21 dah). To identify different typologies of skeletal deformities and quantify their incidence in the three experimental groups 27 specimens per treatment (9 per replicate) were randomly sampled at 66 dah. Fish were anesthetized with clove oil solution (40ppm) and then fixed in 4% formaldehyde in PBS (ph 7.4) for 24 h and preserved in 70% ethanol until further processing. For the visualization of the skeleton and in order to detect normal an abnormal skeletal features, fries were submitted to specific staining procedures that includes alcian blue for cartilage and alizarin red S for calcified structures as described by Gavaia et al (2000). All observations were performed in a fluorescence stereomicroscopy Leica MZ7.5 equipped with a digital camera Olympus F-View. The deformities were separated according to the affected structures as follows: cephalic (1-4), prehemal (5-9), hemal (10-42) and caudal (43-44) vertebra, caudal, dorsal and anal fin and cephalic area.
Results:
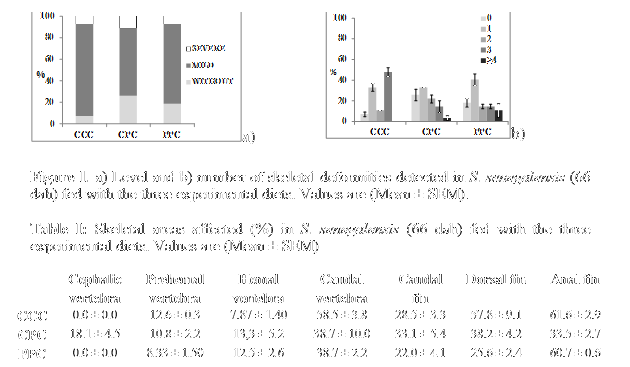
Skeletal deformities registered in the three sole experimental groups were shown in Fig 1(a,b). It was observed a great variability intragroups. No significative differences were detected among diets but the treatment groups revealed a decrease in deformed specimens of up to 11%. Severe deformities were below 12% in the three studied groups and were mainly located in the vertebral column (60%) and the anal fin (40%). No deformities were observed in the cephalic area. The most affected area in the vertebral column was the caudal vertebra (38.7-58.5 % specimens affected) (Table I). Skeletal deformities in cephalic, prehemal and hemal vertebra did not exceed 20% in the three studied groups. Caudal, dorsal and anal fins abnormalities in the treatments ranged 22.0-33.1 %, 25.6-57.8 %, and 33.5-61.6 % respectively (Table I). Probiotic groups had a slightly higher incidence of specimens without deformities (25.9% and 18.5% for CPC and PPC) compared to Control (7.4%) (Figure 1b).In this way Control group had a higher level of multiple abnormalities (48.2%) than probiotic groups (18.5-25.9%).
Discussion and conclusions
Our results indicate a better skeletal condition in sole probiotic groups at the end of the weaning period, but the high variability observed needs further repetitions for robustness. The average rate of deformities was similar to those described by Gavaia et al,(2009) and Boglino et al, (2012). But the level of caudal and anal fin abnormalities was higher than in those studies. The severity of the skeletal deformities detected in this study, involving a loss of less than 12% of the batches, was less than that obtained in sole intensive larviculture by Boglino et al, (2012),. This findings may be due to a slight imbalance of minerals ocurred at larval stages. In conclusion S. putrefaciens Pdp11 bioencapsulated in live diet seems to decrease skeletal deformities in sole fry.Our rearing regime sems to have a positive effect on skeletogenesis during sole larviculture but further studies are needed with higher number of samples to better establish the effects.
Acknowledges:
The present study was supported and financed by MINECO (AGL2011-30381-CO3) and Cantabria Regional Ministry of Cattle Raising, Fisheries and Rural Development. The authors wish to acknowledge for the valuable assistance of IEO staff.
References:
Aubin J. et al., 2005 Aquacult. Res. 36(8): 758-767. Boglino A. et al., , 2012 Aquaculture 324-325: 290-302 Boglione C. et al., 2013 Reviews in Aquaculture 5: S99-S120. Cañavate J.P. and Fernández-Díaz C. 1999. Aquaculture 174: 255-263. Gavaia P.J. et al., 2000 Biotech. Histochem.75(2):79-84 Gavaia P.J. et al., 2009 Aquacult. Res. 40(14): 1585-1593 Lamari F. et al., 2013 Aquaculture 376: 137-145. Lobo et al., 2014 Fish Physiol Bichem. 40(1): 295-309.