DELOUSING WITH HYDROGEN PEROXIDE: SKIN, GILLS AND ESOPHAGUS RESPONSES AND REPAIR IN THE FIRST THREE WEEKS AFTER TREATMENT
Introduction
Infestations by the salmon louse, Lepeophtheirus salmonis cause huge losses to the salmon aquaculture industry amounting to hundreds of millions of Euros annually (Costello, 2009) and is the most important challenge facing industry production and growth today.
Sea lice graze on the skin and mucosal tissue of the fish (Boxaspen, 2006) and this leaves the fish susceptible to secondary infections and osmotic stress, which later leads to mortality (Denholm et al., 2002; Johansen et al., 2011). Hydrogen peroxide (H2O2) is one of several treatments used to control sea lice infection in Atlantic salmon however it may result in an adverse impact to the fish innate immunity
The barrier tissues of fish (skin, gills and guts) have mucosal epithelia which have been shown to play an important role in the defence against pathogens in Atlantic salmon, Salmo salar, (Svendsen and Bogwald, 1997). Mucosal mapping (Pittman et al. 2013) has been shown to allow direct comparison of volumetric data and ratios of epithelia to mucous cell to indicate enhanced immunity, dysregulation in the gut, exposure to infectious agents in the gills and responses to parasites in the skin.
The objective of the work undertaken was to study the effect of hydrogen peroxide treatment on the mucosal barriers of skin, gills and esophagus in commercial salmon farming using mucosal mapping and determine its effect on the salmonids immune system.
Materials and Methods
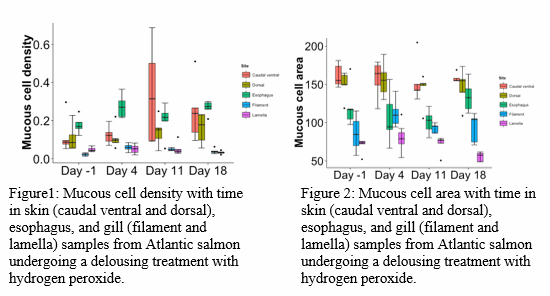
Samples were taken prior to (Day 1), immediately after (Day 4) and at 1 week after (Day 11) and 2 weeks after (Day 18) from Atlantic salmon on a commercial production farm where delousing with hydrogen peroxide was being applied (Day 2). The fish were exposed to a hydrogen peroxide bath at 150g/ L for 20 minutes.
Samples were taken from standardized body sites of dorsolateral skin and ventral caudal peduncle, second gill arch and esophagus, fixed in formalin, decalcified, embedded in either Technovit or paraffin, sectioned, stained with Periodic Acid Schiff and Alcian Blue for unequivocal marking of the mucous cells, and analyzed for mucous cell area and density, mucous cell area:density ratio and filament:lamella ratio for gills using Mucosal Mapping.
Results
Significant differences (p<0.05) in mucous cell density (MCD) (Figure 1) and mucous cell area (MCA) (Figure 2) were always recorded between body sites on the same day. The mean MCA is largest in the skin and smallest in the gills. The MCD of the esophagus is significantly higher on Day 11 and Day 18 relative to before treatment (Day1) (p<0.05). The dorsal MCD was significantly higher only on Day 11 (p<0.05) while the gill lamellae MCD was significantly lower on Day 11 and Day 18 (P<0.05). The ratio of filament epithelium to lamellae epithelium is highest before delousing compared to after delousing while there were no significant differences in filament:lamella epithelium ratio for all days after treatment. The area:density ratio is significantly highest in filament and smallest in esophagus (p<0.001).
Discussion and Conclusion
These results are consistent with previous work on the mucous cell distribution in salmonids. This has shown that the salmonid mucous cell configuration changes as a result of parasites, going from a resting state with medium-sized mucous cells of medium density to a higher density of smaller cells, indicating increased diffusion rates both into and out of these critical cells and more agile motility. This was already the case at day 1.
The gills were significantly affected and weakened after the treatment showing a more sensitive response than the other studied organs. Our results indicate that hydrogen peroxide disrupts the typical architecture of the gills and lesions were observed from moderate (hyperplasia and fused lamellae) to severe (gill epithelial lifting and/or necrosis.
The salmon innate immune system has significant responses to pathogens and to treatments. It is important to take into consideration the H2O2 concentration, treatment exposure time and temperature for delousing. Further work using Mucosal Mapping could be used to determine suitable type and dose treatment for delousing that are least harmful to the salmon and possibly reducing number of chemical treatments required. This can enhance fish welfare and thus reducing the production cost in aquaculture farming.
References
Boxaspen, K. (2006) A review of the biology and genetics of sea lice. ICES Journal of Marine Science: Journal du Conseil, 63 (7):1304-1316.
Costello, M. J. (2009) The global economic cost of sea lice to the salmonid farming industry. Journal of Fish Diseases, 32: 115-118.
Denholm, I., Devine, G. J., Horsberg, T. E., Sevatdal, S., Fallang, A., Nolan, D. V. and Powell, R. (2002), Analysis and management of resistance to chemotherapeutants in salmon lice,Lepeophtheirus salmonis (Copepoda: Caligidae). Pest. Manag. Sci., 58: 528-536.
Johansen, L.H. et al., Johansen, L.-H. , Jensen, I., Mikkelsen, H., Bjorn, P.-A. , Jansen, PA and Bergh, AZ. (2011) Disease interaction and pathogens exchange between wild and farmed fish populations with special reference to Norway. Aquaculture, 315(3-4):167-186.
Pittman K, Pittman A, Karlson S, Cieplinska T, Sourd P, Redmond K, Ravnøy B and Sweetman E. (2013) Body site matters: an evaluation and application of a novel histological methodology on the quantification of mucous cells in the skin of Atlantic salmon Salmo salar L. Journal of Fish Diseases 36(2):115-127
Svendsen Y and Bøgwald J. (1997) Influence of artificial wound and non-intact mucus layer on mortality of Atlantic salmon (Salmo salar L.) following a bath challenge with Vibrio anguillarum and Aeromonas Salmonicida. Fish & Shellfish Immunology 7:317-325