EFFECTS OF DIETARY SUPPLEMENTATION OF Gracilaria sp. EXTRACTS ON GROWTH PERFORMANCE, FILLET QUALITY, OXIDATIVE STRESS AND IMMUNE RESPONSES IN EUROPEAN SEABASS Dicentrarchus labrax
Introduction
By-products generated from the seaweed (SW) industry´s extraction processes have potential applications as feed supplements for aquafeeds. SW extracts contain natural pigments and compounds with antioxidant and immune-stimulant properties [1, 2]. In spite of their beneficial effects, SW extracts may contain compounds with anti-nutritional effects when supplemented in diets, negatively affecting fish growth performance, welfare and ultimately fillet quality [3].
This study appraised the effects of two methanolic fractions extracted from the red SW (Gracilaria sp.), supplemented in European seabass (Dicentrarchus labrax) diets. Three experimental diets were prepared: (i) a control diet without supplementation (CTRL), (ii) a control diet supplemented with the Gracilaria methanolic extract (GE, 0.5%), or (iii) a control diet supplemented with the insoluble residue of the extraction (GR, 4.5%). After 42 days of feeding, the growth performance, digestive enzymes activities, fillet pH and colour, skin colour, and immune and oxidative stress responses were analysed.
Material and Methods
The red SW Gracilaria sp. was produced and supplied by ALGAPLUS (Ílhavo, Portugal). Gracilaria sp. was previously washed, dried and the resulting powder material was percolated with methanol at room temperature until exhaustion. The methanolic solution was evaporated, under reduced pressure, producing a crude methanol extract.
Diets, manufactured by SPAROS Lda. (Olhão, Portugal), were isoproteic (50% crude protein), isolipidic (20% crude lipid) and isoenergetic (23 Kj.g-1 gross energy). Seabass were fed daily at 09:30 and 16:30h for 42 days. The feeding schedule was adjusted daily according to expected growth and detected mortality, with a feeding level set at 2% (DM) of average body weight. Tanks were inspected for left-over feed regularly after feeding. The mortality during the experiment was 3.89% for control, 1.94% for GE and 3.61% for GR. The physical-chemical parameters of the rearing water, such as O2 level (85±10 % saturation), pH (7.9±0.6), temperature (20.4±0.6 ºC) and salinity (30 ppm) were monitored and adjusted during the entire trial. Total ammonium and nitrates maintained at residual levels. The photoperiod was set for 12 h light:12 h dark.
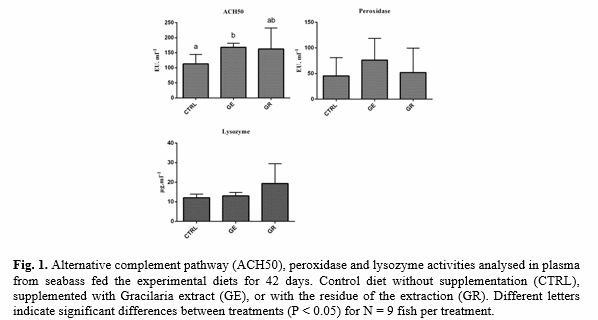
At the end of the feeding trial, fish were weighted and blood, liver and intestine were sampled. Fillets were sampled for colour and pH measurements. Several fish performance parameters were calculated (Weight gain, feed conversion ratio, hepatosomatic index). Specific activity of the digestive enzymes α-amylase, lipase, trypsin and chymotrypsin were measured in whole intestine homogenates. Fillet quality analysis was performed based on pH and flesh colour measurements, once per day for a period of seven consecutive days. Measures of L*, a*, and b* values reflect the human eye response to light and colour perception. Additionally, at day seven, skin colour measurements were performed based on the same methodology as fillet colour. Oxidative stress biomarkers were determined in liver homogenates. Immune status was assessed through the determination of key innate immune parameters, namely lysozyme, peroxidase and the alternative complement pathway levels.
All data was tested for normality (Shapiro-Wilk test) and homogeneity of variances (Levene´s test). The analysis of variance was performed applying One-Way-ANOVA test using the dietary treatments as the dependent variable. Significant differences were considered when P < 0.05. As for fillet characteristics, Mann-Whitney U tests were used for the statistical analysis of the L*, a* and b* colour measurements and ANOVA for differences in pH.
Results
Juvenile seabass grew up to 2.5-fold from initial weight in 42 days of feeding trial. Weight gain (WG: 22.2-24.7 g), daily growth index (DGI: 2.0-2.1% BW day-1), voluntary feed intake (VFI: 1.9-2.0% BW day-1) and feed conversion ratio (FCR: 0.8-0.9), showed no differences between groups, regardless of dietary treatment.
Seabass showed no apparent differences in amylase activity, an indicator of dietary carbohydrate utilization, regardless of the experimental diet provided. In addition, lipase, trypsin and chymotrypsin activities did not differ between the dietary treatments (P > 0.05).
No significant differences were observed in fillet pH and L* value between the dietary groups. Fish fed the GE diet showed significant differences in b* value (P = 0.028) in the posterior L* value. The fillet a* value (P = 0.026) of the anterior measurement differed between GE and CTRL groups. GR group had a* value of posterior measurement significantly different (P = 0.018) from CTRL group on day 3. Skin colour measurements on day 7 revealed significantly higher a* (P = 0.012) and L* (P = 0.020) values in fish fed GE diet when compared with fish fed the CTRL diet. Fish fed GR diet had a significant higher a* (P = 0.035) and showed a tendency of higher L* (P = 0.077) when compared to fish fed CTRL diet.
Lipid peroxidation levels (LPO), catalase (CAT), glutathione peroxidase (GPX), glutathione reductase (GR) and total glutathione (TG) did not vary between the dietary treatments (P < 0.05).
Seabass fed GE diet showed higher alternative complement activity (ACH50) when compared to the CTRL diet (P = 0.042). Plasma peroxidase and lysozyme did not vary among the dietary treatments (Fig. 1).
Discussion
Results revealed that both GE and GR had no apparent negative effects when supplemented into seabass diets, as no differences for growth performance, digestive enzyme activities, fillet pH, and oxidative stress biomarkers were observed after a 42-day feeding trial. Therefore, this study showed that seabass can be fed diets including Gracilaria sp. methanolic extraction products with no negative impacts on zootechnical, biochemical or quality parameters. However, dietary supplementation with SW extraction products produces significant differences in the fillet colour. In addition, changes in the alternative complement pathway may suggest that innate immune parameters may be stimulated by dietary supplementation with SW extraction products.
Acknowledgments
This study has been part of an AQUAEXCEL application for DLO-IMARES research infrastructure with the reference code 0094/07/15/32/B and by Strategic Funding UID/Multi/04423/2013 through national funds provided by FCT - Foundation for Science and Technology and European Regional Development Fund (ERDF), under the framework of the program PT2020.
References
1. Torrecillas S, Makol A, Caballero MJ, Montero D, Robaina L, Real F, et al. Immune stimulation and improved infection resistance in European sea bass (Dicentrarchus labrax) fed mannan oligosaccharides. Fish & shellfish immunology. 2007 23:969-81.
2. Samad R. Effects of dietary supplementation of spirulina and quercetin on growth, innate immune responses, disease resistance against Edwardsiella tarda, and dietary antioxidant capacity in the juvenile olive flounder Paralichthys olivaceus. Fisheries and Aquatic Sciences. 2013 16:7-14.
3. Fleurence J, Levine I. Seaweed in Health and Disease Prevention: Elsevier Science; 2016.