17A-METHYLTESTOSTERONE AND 17B-ESTRADIOL IMPLANT EFFECTS ON THE INDUCTION OF VITELLOGENESIS IN FEMINIZED EUROPEAN SILVER EELS Anguilla anguilla
Introduction
Successful a ssisted propagation of the European eel will lead to a closed production cycle supplying the aquaculture industry with glass eels. The current protocol for the propagation of European eel requires a long period of weekly hormonal injections with pituitary extract (Palstra et al., 2005) which is stressful for the brood stock eels and has negative impact on gamete quality and reproductive success. This procedure can at least partly be replaced by a single injection of a steroid implant (Thomson-Laing et al., 2019). In this study, we have tested the effects of 17α-methyltestosterone (17MT) , as potent androgen activating the androgen receptor (Todo et al., 1999), and 17β-estradiol (E2), as inducer of vitellogenesis.
Materials and methods
F our groups of feminized eels (Chai et al., 2010) were subjected to a simulated migration (Mes et al., 2016) and subsequently injected with implants containing 17MT (T-group) or E2 (E-group); 17MT plus E2 (TE- group) to test the synergistic effect, or without any steroids as controls (C-group). Effects of a 2-month treatment on sexual maturation were investigated by assessment of the eye index (EI), hepatosomatic and gonadosomatic index (HSI and GSI, respectively), plasma steroids as determined by liquid chromatography mass spectrometry (LC-MS ; Blokland et al. , 2017), gonadal histology and rtPCR of androgen receptors a and b (ara , arb); estrogen receptor 1 (esr1); FSH receptor (fshr); vitellogenin receptor (vtgr) and aromatase (cyp19)(Jéhannet et al., 2019).
Results and discussion
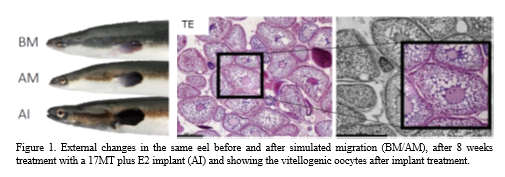
After simulated migration, experimental eels initiated early previtellogenesis as shown by an increase in eye size (Fig. 1) , GSI and oocyte diameter and a lower HSI. P lasma steroid levels also showed marked changes, among others an increase in 11KT and a decrease in E2. For many parameters, both the T and E group showed an increase vs. controls, with the TE group showing an even further increase indicating a synergistic effect: EI, GSI (3.4 for T and for E, 6.6 for TE), oocyte diameter (Fig.1) and ara , arb and esr1 expression. Some parameters reflected 17MT or E2 specific effects. Only eels of the T-group showed increased expression of cyp19 and of fshr , while fshr expression increased 44 fold in the TE group showing that 17MT in combination with E2 is most effective in raising fshr mRNA levels. Specific for eels of the E groups were vitellogenic changes such as an increase of HSI, plasma dehydroepiandrosterone (DHEA) and E2, and the presence of yolk in the oocytes of E and TE (Fig. 1) , not for eels of the T group. O f note are further the effects of time and/or starvation occurring in the controls so independently from 17MT and E2 steroid action after simulated migration. Thus, EI, GSI, oocyte diameter, but also fshr and vtgr expression, increased after simulated migration, and independent of exogenous steroid treatment.
In conclusion, E2 is necessary to start vitellogenesis but 17MT has specific effects on cyp19 and fshr expression. The combination is necessary for the full array of synergistic effects although 17MT implant treatment, followed up in time by E treatment, may result in a more natural sequence of 17MT-induced previtellogenic and E2-induced vitellogenic effects. As such, steroid implants could be applied in assisted reproduction protocols for European eel as slow release systems to initiate the vitellogenesis and to reduce the number of weekly hormonal injections, potentially improving oocyte quality and leading to production of more viable and robust larvae.
Acknowledgements: The authors thank the DUPAN foundation; The Dutch Ministry of Economic Affairs and the European Union, European Maritime and Fisheries Fund, and partners of the international EELRIC consortium (www.eelric.eu).
References
Blokland MH, Van Tricht EF, Van Ginkel LA, Sterk SS., 2017. Applicability of an innovative steroid-profiling method to determine synthetic growth promoter abuse in cattle. J Steroid Biochem Mol Biol 2017; 174:265–275.
Chai Y., Tosaka R., Abe T., Sago K., Sago Y., Hatanaka E., Ijiri S. , Adachi S., 2010. The relationship between the developmental stage of oocytes in various seasons and the quality of the egg obtained by artificial maturation in the feminized Japanese eel Anguilla japonica . Aquaculture Sci. 58, 269-278.
Jéhannet , P., Kruijt, L., Damsteegt, E.L., Swinkels, W., Heinsbroek, L.T.N., Lokman, P.M., Palstra, A.P., 2019. A mechanistic model for studying the initiation of anguillid vitellogenesis by comparing the European eel (Anguilla anguilla) and the shortfinned eel (A. australis). Gen Comp Endocrinol 279, 129-138.
Mes, D., Dirks, R.P., Palstra, A.P., 2016. Simulated migration under mimicked photothermal conditions enhances sexual maturation of farmed European eel (Anguilla anguilla ). Aquaculture 452, 367–372.
Palstra, A.P., Cohen, E., Niemantsverdriet, P., van Ginneken, V., van den Thillart, G.E.E.J.M., 2005. Artificial maturation and reproduction of European silver eel: Development of oocytes during final maturation. Aquaculture 249: 533-547
Thomson-Laing, G., Damsteegt, E.L., Nagata, J., Ijiri, S., Adachi, S., Todo, T., Hiramatsu, N., Lokman, P.M., 2019. Synergistic effects of estradiol and 11-ketotestosterone on vitellogenin physiology in the shortfinned eel (Anguilla australis ). Biology of Reproduction 100, 1319–1332.
Todo, T., Ikeuchi, T., Kobayashi, T., Nagahama, Y., 1999. Fish Androgen Receptor: cDNA Cloning, Steroid Activation of Transcription in Transfected Mammalian Cells, and Tissue mRNA Levels . Biochemical and Biophysical Research Communications 254, 378-383.