CHARACTERIZATION OF MOLECULAR, CELLULAR AND HUMORAL IMMUNE MARKERS IN THE EUROPEAN SEABASS Dicentrarchus labrax UNDER CHRONIC INFLAMMATORY CONDITIONS
Introduction
Fish health and welfare are among the top priorities in what aquaculture research is concerned. Stress-inducing husbandry and rearing conditions (including alternative dietary formulations), bacterial infections or parasitic diseases may all lead to chronic inflammation (1, 2). Such an immune response channels energy away from growth, reproduction and other important physiological processes, to fuel immune-related metabolic responses. Despite a reasonable amount of studies on fish immune response, there is a lack of a solid inflammation model for fish and a battery of biological markers that can more accurately characterize chronic inflammatory responses. The present study aims to gather information on molecular, cellular and humoral parameters of European seabass (Dicentrarchus labrax) undergoing chronic inflammation that can be used as health indicators for applications in fish health management.
Material and methods
Juvenile European seabass (300 g ± 71.8) were acclimatized in a recirculating seawater system (temperature 16 ºC; Salinity: 35; Photoperiod: natural summer time; n = 12) for 30 days being fed a commercial diet throughout the whole experiment. At the end of this period, fish were intra-peritoneally injected with either Freund’s Incomplete Adjuvant (FIA) to induce inflammation or Hanks Balanced Salt Solution (HBSS) to serve as sham, and distributed in duplicate tanks in the same system. Fish were sacrificed at 24 h, 7 days, 14 days and 21 days post-injection (n = 3 per tank). During sampling, blood was collected from the caudal vein and directly used for total peripheral cell counts, blood smears preparation and respiratoy burst. Plasma was then obtained for the assessment of humoral immune parameters. Peritoneal exudates were collected for total peritoneal leucocytes counts. The head-kidney was sampled for the evaluation of immune-related and opioid receptors gene expression measured by real-time quantitative PCR.
Results
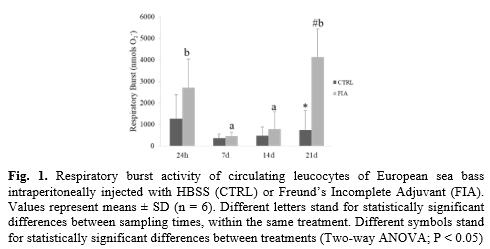
Though there were no significant differences in total peripheral cell counts between FIA and HBSS groups, FIA-injected fish presented higher total peritoneal leucocyte concentration than the HBSS group at 7, 14 and 21 days post-injection. Peripheral monocytes were also higher in FIA-injected fish than in the HBSS group regardless of sampling time, as well as both lymphocytes and neutrophils at 24h post injection. Total proteases activity and lysozyme activity were also induced in FIA-injected group, compared to the HBSS group, regardless of sampling time. Similarly, increased respiratory burst was observed in FIA-injected fish, though only in fish sampled at 21 days post-injection (Fig 1). Regarding head-kidney gene expression, interleukin 1β (il1β) and caspase 1 (casp1) were both up-regulated in FIA-injected fish, irrespective of sampling time. Moreover, interleukin 34 (il34) did not respond differently between groups or sampling times, but an increasing trend was observed within FIA-injected fish. In what opioid receptors are concerned, no differences were detected between FIA and HBSS groups but mu, kappa and nociceptin opioid receptor (muor, kor1 and nopr, respectively) were found down-regulated in fish sampled at 7 d, 14 d and 21 d post-injection compared to levels measured at 24 h.
Discussion and conclusion
FIA is well-known to trigger and enhance inflammatory responses, thus serving as a very effective adjuvant during vaccination. Indeed, the results here reported are clear indicators of an inflamed peritoneal cavity and an ongoing systemic immune response that persist at least for 21 days. Locally, inflammation was characterized by an intense recruitment of immune cells that was still evident 21 days after injection thus illustrating the chronic character of the immune response. Cellular response was also noticed peripherally with neutrophils, lymphocytes and monocytes numbers rising in the blood of FIA-injected fish. Furthermore, cellular-mediated respiratory burst peaked at 21 days post FIA injection, suggesting that phagocytes were still actively fighting the phlogistic agent. Respiratory burst is a particularly interesting parameter to be seen enhanced at such a late time-point, as it is a key discriminant feature of active phagocytes (3), very much present at the onset of inflammation. Regarding the head-kidney molecular analysis, pro-inflammatory genes (il1β, casp1) were indeed higher in fish injected with FIA but such effect was regardless of sampling point, making these poor markers of a prolonged response. il34 expression time-dependent increasing trend poses as good marker of chronic inflammation due to its importance as a second ligand of CSFR1, a pathway with high relevance in chronic inflammation settings (4). In conclusion, peripheral leucocyte counts and respiratory burst analysis seem to be reliable indicators of chronic inflammation easily obtained with a blood sample. Further investigation is nonetheless required to select more responsive and specific molecular markers.
Acknowledgements
This work was supported by project INFLAMMAA (PTDC/CVT-CVT/32349/2017), financed by Portugal and the European Union through FEDER, COMPETE 2020 and CRESC Algarve 2020, in the framework of Portugal 2020, and through national funds through Fundação para a Ciência e a Tecnologia (FCT, Portugal). BC and DP were also supported by FCT (IF/00197/2015 and UI/BD/150900/2021, respectively).
References
1. Hedrera et al. 2013. DOI: 10.1371/journal.pone.0069983
2. Dezfuli et al. 2008. DOI: 10.1051/parasite/2008153426
3. Plouffe et al. 2005. DOI: 10.1111/j.1399-3089.2005.00227.x
4. Masteller and Wong. 2014 DOI: 10.1016/j.drudis.2014.05.016