LOW SPERM TO EGG RATIO FOR IN VITRO FERTILISATION OFFERS A SOLUTION FOR THE REPRODUCTIVE CONTROL OF CULTURED SOLE Solea senegalensis
Introduction
Cultured Senegalese sole (Solea senegalensis ) reared from egg to adult in captivity do not spawn spontaneously. Implementation of in vitro fertilisation procedures would appear to be a solution to this problem ( Rasines et al., 2012). However, cultured sole produce small amounts of poor-quality sperm ( González-López et al., 2020) that have been considered inadequate for in vitro fertilisation procedures. The sperm to egg ratio in fish has been observed to vary from 1,000s to 100,000s of sperm per egg (Beirão et al., 2019) , with flatfish generally exhibiting the lowest sperm to egg ratios. The present study aimed to de fine the sperm to egg ratio required to achieve high levels of fertilisation o n an experimental scale and using commercially relevant numbers of eggs.
Materials and Methods
The cultured Senegalese sole used were hatched and reared entirely in captivity (females of 1.53 ± 0.28 kg and males of 1.05 ± 0.25 kg) . Fish were maintained in 10,000 L tanks in RAS (IRTAmar®) and fed a diet of polychaetes, mussels and pellets (Broodfeedlean, SPAROS) . Photoperiod was natural and water temperature was constant 16 ± 1 ºC. Eggs were obtained by inducing ovulation by administering 5 µg/ kg of GnRHa to females with mean oocyte diameter ≥ 600 µm. The females were held in total darkness until ovulation. Sperm was stripped from untreated culture males (González-López et al., 2020). Sperm concentration was measured (Thoma chamber) and motility assessed with ImageJ CASA . In a sperm to egg ratio experiment, sperm (n=5 males) was serially diluted with modified Leibovitz to obtain eight dilutions: 1:4; 1:19; 1:79; 1:319; 1:959; 1:2879; 1:5759; 1:11519 and triplicate fertilisations of 0.5 mL of eggs (n=5 females) were completed . After 24 hours of incubation (16ºC) the development of ≥ 50 eggs were determined per replica . In a proof of concept experiment > 100,000 eggs were fertilised with ≥ 150 µL of motile sperm in female – male pairs (n=7) . Eggs were incubated and hatching rate assessed. In an egg viability experiment, three batches of eg gs were stored at room temperature and fertilised at collection and at 30 - 60 minute intervals after collection. Egg development was assessed after 24 h. In a sperm collection experiment, the sperm quality (n = 13 males ) was assessed when collected directly into a syringe containing Leibovitz (1:4), diluted in Leibovitz (1:4) 4 ± 2 minutes after collection and undiluted sperm.
Results and Discussion
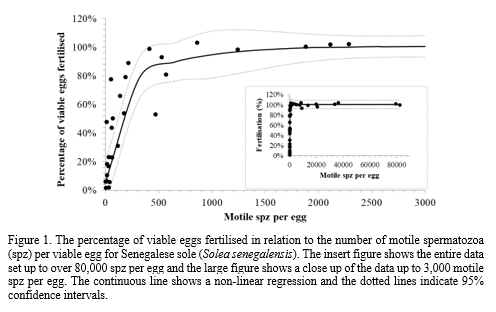
Twenty eight males (60.9 % of 46 checked ) had adequate sperm quantity and quality for the experiments. Eight (62% of 13 induced ) females ovulated good quality eggs (82.6 ± 9.2 % fertilisation) that were used in the experiments. The mean time from GnRHa administration to ovulation was 41:57 ± 1:46 h, mean fecundity was 130,789 ± 36,723 eggs/fish or 87,174 ± 24,378 eggs/ kg of body weight. In the sperm to egg ratio experiment the relationship between percentage fertilisation and sperm per egg was described by a non-linear regression based on an equation for an exponential rise to a maximum with double, five parameters (R = 0.93, P < 0.0001) (Fig. 1). O nly 649 motile sperm fertilised 90 ± 13% (± 95% CI) of viable eggs and 1617 motile spz fertilised 99 ± 12% (± 95% CI) of viable eggs. In addition, percentage motility was negatively correlated to the number of sperm required to fertilise a viable egg (R = -0.93). However, there was no correlation between motile sperm required and percentage of viable eggs. In t he proof-of-concept experiment the mean percentage hatch was 70 ± 14 % to produce 131,540 ± 34,448 larvae per fertilisation. The sperm from a single cultured male , mean volume of 145 ± 50 µL (8 ± 6.8 × 108 spermatozoa) was used to fertilise 190,512 ± 38,471 eggs, which gave a ratio of 592 ± 611 motile sperm per egg. The e gg viability experiment indicated that the percentage of fertilised eggs gradually decreased with storage time with 81 ± 26% (± 95% CI) fertilisation after 30 minutes and 57 ± 20% (± 95% CI) after an hour. The sperm extraction experiment demonstrated that t he percentage motility was significantly (P < 0.05) higher in samples collected directly into modified Leibovitz (33.4 ± 3.5% ) compared to dilution 4 ± 2 minutes after collection ( 6.6 ± 1.6%) or undiluted sperm ( 2.9 ± 1.2%).
Senegalese sole was amongst the lowest sperm to egg ratio recorded in fish (Beirão et al., 2019). The possible relationship between low sperm to egg ratio and sole reproductive characteristics such as paired-spawning , gamete fertilisation in close proximity with no sperm competition will be discussed . Eggs should be fertilised as soon as possible after ovulation and sperm quality was improved with early dilution in modified Leibovitz. The low sperm to egg ratio was tested and shown to fertilise large amounts of eggs (>100,000) with the sperm from a single cultured male ( approx., 200 µL of sperm per 100 mL of eggs) .
Acknowledgements: The authors thank IRTA staff and Josep Lluis Celades and Ignacio Giménez (RARA-AVIS SL), Mario Villalta Vega ( IES Alfacs ) and Alex Rullo Reverté ( IES Alfacs) for technical assistance. The study was funded by INIA-FEDER project nº RTA2014-0048 and supported by the project 038433_REARLING, Portugal and the European Union through ERDF, COMPETE 2020, Portugal 2020, coordinated by Isidro Blanquet (Sea8 Group, Portugal ). PhD grants funded the p articipation of WGL (CONACYT, Mexico) and SRJ (AGAUR , Catalonia).
References
Beirão J, Boulais M, Gallego V, O’Brien JK , Peixoto S, Robeck TR , Cabrita E. 2019 Sperm handling in aquatic animals for artificial reproduction . Theriogenology 133, 161–178. (doi:10.1016/j.theriogenology.2019.05.004)
González-López WÁ, Ramos-Júdez S, Giménez I, Duncan NJ. 2020 Sperm contamination by urine in Senegalese sole (Solea senegalensis) and the use of extender solutions for short-term chilled storage. Aquaculture 516, 734649. (doi:10.1016/j.aquaculture.2019.734649)
Rasines, I., Gómez, M., Martín, I., Rodríguez, C., Mañanós, E., Chereguini, O., 2012. Artificial fertilization of Senegalese sole (Solea senegalensis): hormone administration methods, timing of ovulation and vi ability of eggs retained in the ovarian cavity. Aquaculture 326–329: 129–135.