EFFECT OF DIETARY SUPPLEMENTATION OF MICROALGAE EXTRACTS ON THE SKELETAL HEALTH OF ZEBRAFISH Danio rerio AND GILTHEAD SEABREAM Sparus aurata LARVAL STAGES: A COMPARATIVE STUDY
Introduction
S keletal anomalies are omnipresent in most farmed fishes worldwide , raising concerns for animal welfare and causing economic losses for the aquaculture industry
. L arval n utrition has been recognized as one of the main factors leading to skeletal anomalies
, thus the development of feeds supplemented with osteo-active compounds is seen as an important strategy to improve the skeletal status of farmed fish. In this context, extracts from m arine organisms , a well-established and valuable source of nutrients, have recently been found to contain bioactive compounds with osteogenic and mineralogenic activities that could be highly relevant to improve skeletal health in larvae of commercial species. Here, w e produced inert diets enriched with different concentrations of the ethanolic extracts from Skeletonema sp. and Tetraselmis sp. and tested them on larvae of the model species zebrafish (Danio rerio ) and the commercial species gilthead seabream (Sparus aurata), evaluating their effect on growth, survival and development of skeletal anomalies.
Materials and Methods
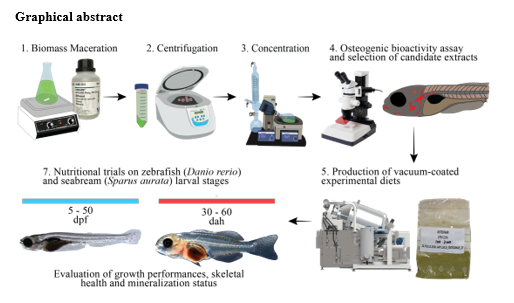
Extracts were prepared through the maceration of f reeze-dried biomass of Skeletonema sp. and Tetraselmis sp. (Necton S.A.) with 96% ethanol . Ethanol extracts were coated on a commercial zebrafish diet (Sparos Lda) at 0.5% and 2.5% and a commercial seabream diet at 0.5% . For zebrafish feeding trials , 5 days post-fertilization (dpf) larvae were maintained in 2 .5 L tanks in static conditions at 60 larvae/L until 20 dpf , then moved to 3 L tanks in recirculating system at a density of 23 larvae/L . Experiments were conducted in triplicates for each of the 5 diets. From 5 to 16 dpf, zebrafish larvae were co-fed with the supplemented inert diet and rotifers , gradually reducing the concentration of rotifers to address the weaning from live feeds. F rom 17 dpf until the end of the trials, larvae were fed only supplemented inert diets . At 50 dpf, juvenile fish were given a lethal anaesthesia and f inal survival, total length and dry weight were assessed. Fish were sampled to asses c alcium and phosphorus content and for double staining with alizarin red S and alcian blue for bony and cartilaginous structures to evaluate skeletal anomalies. T he expression of marker genes of bone development, matrix mineralization and oxidative stress were assessed by qPCR.
For the seabream trials, larvae of 30 days after hatching (dah) were fed for 30 days with control diet or diets supplemented with 0.5% of Tetraselmis sp. or Skeletonema sp. ethanolic extracts. M icrodiets were formulated and processed to be isonitrogenous, and isoenergetic. Larvae were maintained in cylindroconical 100 L tanks in a semi-closed R AS with an initial density of 52 larvae/L and a p hotoperiod of 10h light:1 4h dark. E nvironmental parameters and mortality were monitored daily . At the end of the trial larvae were euthanized, sampled and growth parameters assessed - dry w eight, length and condition factor. Samples for oxidative status, digestive capacity, total mineral content, calcium and phosphorous ratio, mRNA levels of marker genes for bone formation, matrix mineralization, and oxidative stress were collected. Some fish per condition were stained to reveal bone and cartilaginous structures and asses incidence of skeletal anomalies
Results and Discussion
In the past years, our laboratory has screened a large number of extracts from microalgae for bone anabolic activity using zebrafish in vivo tools and identified ethanolic extracts of Skeletonema sp. and Tetraselmis sp. as promising source of osteoactive compounds. Although the use of zebrafish as model organism in aquaculture is a controversial topic due to physiological differences and evolutionary distances between this small freshwater cyprinid and commercial teleost species, its use in nutritional research has been recognized as a great opportunity and r esults obtained in zebrafish were shown to have predictive power of the effect on stress, immunological response and dietary toxicity towards species reared in aquaculture
. Aiming at providing a comparative view on the potential application of microalgae extracts in fin fish nutrition, we produced inert diets enriched with different concentrations of two promising osteogenic extracts , tested them on larval stages of zebrafish and the gilthead seabream, evaluating their effect on growth, survival and the occurrence of skeletal anomalies . Regarding the feeding trials with seabream, results showed that dietary treatments had no impact on performance indicators like dry weight, length, or condition factor . For both species, the impact of the supplementation of the extracts on bone mineral content, expression of marker genes of bone growth and mineralization and incidence of skeletal anomalies revealed the advantages of microalgae extracts supplemented diets, and their potential for aquaculture applications.
Funding
W ork financed by the European Maritime and Fisheries Fund (EMFF/FEAMP) through the National Operational Programme MAR2020 (grant 16-02-01-FMP-0057/OSTEOMAR) , by the European Regional Development Fund (ERDF/FEDER) through the Transnational Cooperation Programme Atlantic Area (grant EAPA/151/2016/BLUEHUMAN ), by the Marie Skłodowska-Curie innovative training network BIOMEDAQU ( grant H2020-MSCA-ITN /766347) and by National funds through the Portuguese Foundation for Science and Technology (grant UIDB/04326/2020).
Bibliography
1. Boglione, C., Gavaia, P., Koumoundouros, G., Gisbert, E., Moren, M., Fontagné, S., and Witten, P. E. (2013) Skeletal anomalies in reared European fish larvae and juveniles. Part 1: Normal and anomalous skeletogenic processes. Rev. Aquac. 5, 99–120
2. Boglione, C., Gisbert, E., Gavaia, P., Witten, P. E., Moren, M., Fontagné, S., and Koumoundouros, G. (2013) Skeletal anomalies in reared European fish larvae and juveniles. Part 2: Main typologies, occurrences and causative factors. Rev. Aquac. 5, 121–167
3. Rønnestad, I., Yúfera, M., Ueberschär, B., Ribeiro, L., Sæle, Ø., and Boglione, C. (2013) Feeding behaviour and digestive physiology in larval fish: Current knowledge, and gaps and bottlenecks in research. Rev. Aquac. 5, S59-S98.
4. Hamre, K., Yúfera, M., Rønnestad, I., Boglione, C., Conceição, L. E. C., and Izquierdo, M. (2013) Fish larval nutrition and feed formulation: Knowledge gaps and bottlenecks for advances in larval rearing. Rev. Aquac. 5, S26-S58.
5. Ribas, L., and Piferrer, F. (2014) The zebrafish (Danio rerio) as a model organism, with emphasis on applications for finfish aquaculture research. Rev. Aquac. 6, 209–240
6. Ulloa, P. E., Medrano, J. F., and Feijo, C. G. (2014) Zebrafish as animal model for aquaculture nutrition research. Front. Genet. 5, 1–6