OLIVE BIOACTIVES AS A CIRCULAR BIOECONOMY SOLUTION TO INTESTINAL INFLAMMATION IN EUROPEAN SEABASS Dicentrarchus labrax
Introduction
Inflammation in aquaculture species can arise from various stressors such as suboptimal diets, environmental fluctuations, and pathogen exposure. Chronic or unresolved intestinal inflammation impairs nutrient absorption, compromises immune function, and ultimately hinders growth performance and overall fish welfare. Exploring functional ingredients with anti-inflammatory properties offers a promising strategy to support intestinal integrity and enhance resilience in farmed fish. Spain, by far the world’s largest olive oil producer, generates large volumes of by-products, including olive pomace containing valuable bioactives like triterpenoids. Therefore, olive-derived by-products have great potential to offer a circular, eco-friendly approach to mitigate inflammation, and enhance productivity and animal well-being, improving overall aquaculture sustainability. To test this hypothesis, we used a protocol developed by Sparos Lta. to chemically induce intestinal inflammation in European seabass using dextran sodium sulphate (DSS).
Materials and methods
The trial comprised 2 phases. In phase I, the whole fish population was fed a diet containing DSS for 15 days to induce a leaky gut and inflammatory response. Then, six fish were sampled to assess the intestinal inflammatory status, and the remaining were sorted and homogeneously distributed into quadruplicate 500L tanks, with 45 fish/tank, supplied with recirculated and aerated water. Phase II consisted in hand feeding the fish (start weight: 35.0±2.1 g) the experimental treatments to visual satiety, 2-3 times per day, for 61 days. A control diet (CTRL) was formulated mimicking a practical low fishmeal diet, with 44% crude protein, 17% crude fat level and 21.3 MJ/kg gross energy. Two additional treatments were prepared by supplementing the same formulation with 0.2% or 0.4% of an additive containing olive pomace extract (OE). Water quality was monitored throughout the trial and kept at good levels; average temperature was 19.5±0.3ºC. At the end of phase II (day 77), all fish were weighed, 6 fish/tank were pooled for whole-body composition analysis, and 3 individual fish/tank were sampled for subsequent analysis, to assess growth, feed utilization and intestinal health status (recovery from inflammation).
Results and discussion
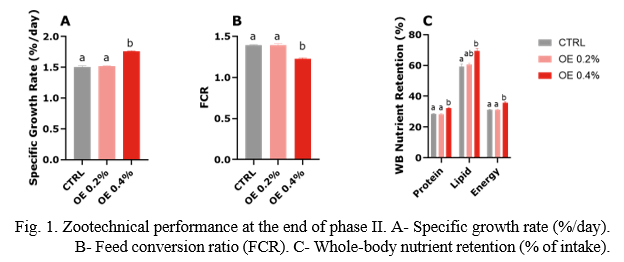
At the end of phase II, fish reached, on average, 87.6g, 88.2g or 102.5g in the CTR, OE 0.2% or OE 0.4% treatments, respectively. This represented a significant 17% higher growth in fish fed OE 0.4% during the recovery period. Feed intake was not affected by OE dietary inclusion. As a result, FCR was also significantly improved (by 11.7%) in fish fed the OE 0.4% diet. Moreover, this treatment had a higher whole-body retention (% of intake) of protein, fat and energy (Fig. 1).
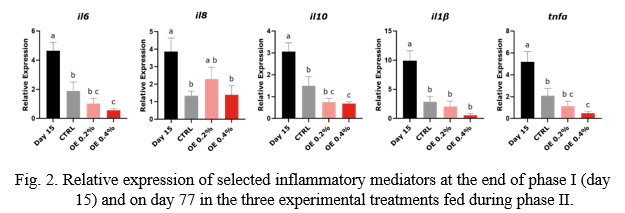
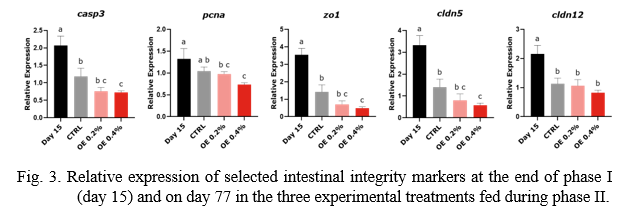
Intestinal gene expression was analyzed by RT-qPCR in the general fish population on day 15 (end of the DSS administration period), and on day 77 in each treatment to verify the degree of recovery from intestinal inflammation. A panel of genes was selected to evaluate the inflammatory response (Fig. 2) and integrity (Fig. 3) of the intestinal epithelium. Results were normalized by the average expression of ef1a, bact and gapdh.
All results shown below correspond to the mean ± SEM. Bars with different letters denote significant differences, assessed by one-way ANOVA and Fisher’s LSD (P<0.05).
Intestinal expression of inflammatory mediators (il6, il8, il10, il1β, tnfɑ and nfkb2) was significantly reduced between day 15 and day 77, indicating that fish were able to at least partly recover from inflammation by the end of phase II (Fig. 2). Additionally, the expression of il6, il10 and tnfɑ was significantly lower in OE 0.4% than in the CTRL on day 77, suggesting that the lowest inflammatory status was achieved in this treatment.
Markers of apoptosis (casp3) and cellular proliferation (pcna), and genes coding for tight junction proteins (zo1, cldn5 and cldn12) were significantly elevated on day 15 (Fig. 3). This suggests that a compensatory mechanism was activated to counteract the loss of intestinal barrier integrity. Conversely, the significantly lower expression of these genes (except cldn12) in the OE 0.4% treatment, relative to the CTRL, could be a sign of improved membrane integrity and higher recovery from DSS-induced damage.
These results clearly indicate a positive effect of the OE at 0.4% dietary inclusion level in alleviating intestinal inflammation in European seabass. Moreover, the faster and/or higher recovery from the DSS-induced disruption of intestinal integrity and inflammation was associated with significant improvements in fish growth, feeding efficiency and whole-body nutrient retention. Results are still pending on histological examination of the intestine, and plasma and feces inflammation markers, which are expected to further support the potential of triterpene-rich OE as a practical solution for mitigating intestinal inflammation challenges in fish farming.