COMPARATIVE ANALYSIS OF THE CREATINE TRANSPORTER SLC6A8 IN RAINBOW TROUT Oncorhynchus mykiss: INSIGHTS INTO VARIANT-SPECIFIC EXPRESSION AND FUNCTION IN TELEOST FISH
Introduction
The creatine system in vertebrates is an important cellular energy buffer , maintaining high-energy phosphate bonds that quickly replenish adenosine triphosphate (ATP) when needed. This is especially important in tissues with a high energy demand like the brain, heart or muscle . Creatine can be gained via nutrition or it can be synthesized intrinsically through a two-step mechanism catalyzed by the enzymes glycine amidinotransferase (GATM), guanidinoacetate N-methyltransferase (GAMT) and creatine kinases (CKs). The uptake of creatine from the bloodstream into cells is mediated by the sodium- and chloride dependent creatine transporter 1 (CT1) , which belongs to the solute carrier 6 (SLC6) transport family of secondary active Na+/Cl- co-transporter (Kristensen et al., 2011).
Due to their different habitats, fishes have developed different metabolic strategies compared to other vertebrates. We have recently shown that teleost fish and mammals differ in regard to their creatine systems. The findings indicate the presence of a muscle-specific creatine synthesis in fish (Borchel et al., 2019). Based on these differences, the present study was aimed to further enhance the knowledge on the transportation of creatine in a teleost model species . Hence, we characterized the SLC6A8 genes encoding the creatine transporter CT1 in rainbow trout Oncorhynchus mykiss. Structural and functional properties in terms of evolutionary conservation, as well as their role as creatine transporter in a salmonid cell model were investigated by complementary molecular techniques.
Material & Methods
Database searches identified two entries in the NCBI (National Center for Biotechnology Information; http://genomicus.biologie.ens.fr/genomicus) and the construction of a phylogenetic tree for the SLC6 family in MEGA 11 software by using the Neighbour-Joining method including 1000 bootstrap replicates and the Poisson correction method.
Expression of creatin system genes SLC6A8a , -b , GATM , GAMT , and CKM in rainbow trout was quantified in different tissues and the salmonid cell line CHSE-14 (Sigma-Aldrich) by q uantitative PCR on the LightCycler96 system (Roche Diagnostics) and in publicly available RNA-Seq data from BioProject PRJNA638521.
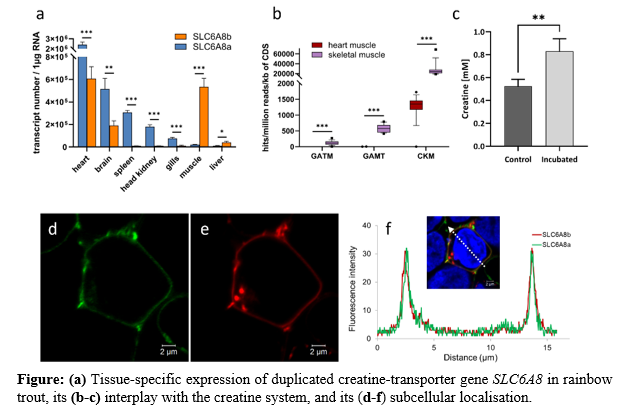
Live-cell imaging was performed using the confocal microscopy LSM 780 (Carl Zeiss Microscopy) to determine the subcellular localisation of constructed GFP-SLC6A8a - and Plum-SLC6A8b or GFP-SLC6A8a and Plum-SLC6A6 fusion genes. Creatine uptake in pre-incubated CHSE-214 cells was determination in extracts of white-muscle tissue.
Results and Discussion
The SLC6A8a and SLC6A8b gene variants show variant-specific expression patterns (Figure a) . Both exhibit the highest transcript level in the heart. Despite this similarity, SLC6A8a significantly exceeds the expression of SLC6A8b in the heart, brain, spleen, head kidney and gill tissue, while SLC6A8b is predominantly expressed in the muscle and liver. Moreover, w e detected clear differences in the expression of the central creatine system genes GATM , GAMT and CKM in the heart and skeletal muscle (Figure b) suggesting an overall different creatine metabolism between both tissues in rainbow trout.
The pre-incubation of the CHSE cells with creatine resulted in its significant increase in concentration in the cells from 0.52 mM to 0.83 mM (Figure c) , indicating its carrier-dependent transport of creatine across the cell membrane . In line with this, we verified the cellular localisation of rainbow trout proteins SLC6A8a and SLC6A8b in the cell membrane (Figure d-f).
The present study extends the knowledge of the creatine transporter SLC6A8 in rainbow trout and supports the previously reported differences in the creatine system of mammals and teleost fishes. However, further functional analyses are required to deepen the knowledge on the variant-specific membrane transport of creatine by SLC6A8a and SLC6A8b in fish.
Key references
- Kristensen, A.S., Andersen, J., Jorgensen , T.N., Sorensen, L., Eriksen, J., Loland , C.J., Stromgaard , K., Gether , U., 2011. SLC6 neurotransmitter transporters: Structure, function, and regulation. Pharmacol . Rev. 63, 585–640. https://doi.org/10.1124/pr.108.000869
- Borchel , A., Verleih, M., Kühn, C., Rebl, A., Goldammer, T., 2019. Evolutionary expression differences of creatine synthesis-related genes: Implications for skeletal muscle metabolism in fish. Sci. Rep. 9, 5429. https://doi.org/10.1038/s41598-019-41907-6