DEMAND COVERAGE IS NOT ENOUGH: THE INFLUENCE OF VARYING LYS TO MET + CYS RATIOS ON FEED INTAKE IN RAINBOW TROUT Oncorhynchus mykiss
Introduction
Fishmeal is still one of the main protein sources in fish feed, but its increasing demand can only be covered by sustainable aquaculture that uses alternative protein sources (FAO, 2022; Hua et al., 2019; Sarker et al., 2020). Therefore, in this project, raw materials that were suitable for organic production were used. Altho ugh plant-based protein sources have been tested as substitutes for fishmeal, these often have negative effects on digestion, taste, and growth due to a suboptimal amino acid composition (de Francesco et al., 2004; Francis et al., 2001; Lazzarotto et al., 2018; Kaiser, 2021; Nagel et al., 2012; Slawski, 2012) . There are also indications that an amino acid imbalance could reduce feed intake (Bak et al., 2012; Comesaña et al., 2017; Comesaña et al., 2020 ). The purpose of this study was to test raw materials as fishmeal substitutes to investigate the influence of varying Lys to Met+ Cys ratios above demand, because it is assumed that these higher ratios can positively affect feed intake and growth.
Material and Methods
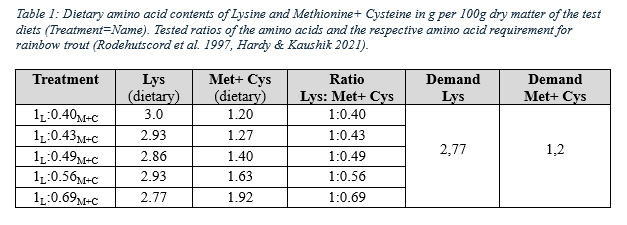
Six different diets were formulated, which varied in terms of raw material and amino acid composition in terms of Lys to Met+ Cys ratios. The control diet included fishmeal and soy protein as the main protein sources. In five experimental diets, the fishmeal and soy protein were completely replaced by different combinations of pea protein, poultry meal, soldier fly larvae meal, and feather meal, resulting in different dietary amino acid compositions in terms of Lys to Met+ Cys ratios (Table 1). All diets were isonitrogenous and isoenergetic at the digestible level and were tested in triplicates. Rainbow trout with an initial weight of ~26g were held in a recirculation aquaculture system and fed daily to apparent satiation for 52 days.
Results and Discussion
The group fed the diet with a lysine to methionine+ cysteine ratio of 1:0.40 showed a significantly higher DFI (3.11±0.06) than the overall mean. Fish fed the diet with a ratio of 1L:0.49M+C showed a significantly lower FCR (1.05±0.03). Both diets showed a higher SGR (2.86±0.03). Both treatments contained the highest levels of pea protein (25.1%; 17.4%), which, according to Gatlin III et al. (2007), is suitable as a moderate fishmeal substitute as a high proportion of lysine and methionine is provided. The amino acid composition might affect the higher feed intake of the diet 1L:0.40M+C. This diet contained the highest levels of isoleucine, leucine and arginine. According to Yamashita, Yamada & Hara (2005), isoleucine and leucine have a positive effect on feed intake in rainbow trout. However, Comesaña et al. (2020) showed a negative effect on the provision of leucine on feed intake. Arginine is an effective taste stimulant in fish, but its influence is negligible due to the husbandry conditions prevailing in this experiment, as their receptors are active only at a pH of 8.5 (Marui et al., 1983). The SGR and FCR values of the diet 1L:0.49M+C could also be attributed to the amino acid composition, with a methionine and cysteine ratio of 1:1, which may benefit starch and fat digestion (Nordrum et al.,1999). Some studies showed that cysteine might be a positive stimulant for locomotory activity in rainbow trout and feed intake (Yamashita, Yamada & Hara, 2005; Kasumyan & Doving, 2003).
Conclusion
The results show that organically produced raw materials in fish nutrition can offer sustainable alternatives to conventionally produced feeds. It could also be demonstrated that feed formulations with different ratios of lysine to methionine+ cysteine significantly influence feed intake, feed conversion and specific growth rate in fish. In addition, the ratio of methionine to cysteine appears to influence nutrient digestion, which could improve growth performance. Follow-up experiments examine more closely the optimal balance of these amino acids and the potential of ecological resources to maximise feed efficiency and growth in aquaculture.
References
Bak, L. K., Johansen, M. L., Schousboe, A. & Waagepetersen, H. S. (2012): Valine but not Leucine or Isoleucine supports Neuro-transmitter glutamate synthesis during synaptic activity in cultured cerebellar neurons, Journal of neuroscience research, 90: 1768-1775.
Comesaña, S., Velasco, C., Ceinos, R. M., López-Patiño, M. A., Míguez J. M., Morais, S. & Soengas, J. L. (2017): Evidence for the presence in rainbow trout brain of amino acid- sensing system involved in the control of food intake. AJP-Regul Integr Comp Physiol, 314: R201-R215.
Comesaña, S., Conde-Sieira, M., Velasco, C., Soengas, J. L., Morai, S. (2020): Oral and pre-absoptive sensing of amino acids relates hypothalamic control of food intake in rainbow trout. Journal of Experimental Biology, 223.
de Francesco, M, Parisi, G., Médale, F., Lupi, P, Kaushik, S. J., & Poli, B. M. (2004): Effect of long-term feeding with a plant pro-tein mixture-based diet on growth and body/fillet quality traits of large rainbow trout (Oncorhynchus mykiss). Aquaculture, 236(1-4): 413-429.
Francis, G., Makkar, H. P, & Becker, K. (2001) : Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture, 199(3-4): 197-227.
FAO. The State of World Fisheries and Aquaculture 2022: Sustainability in Action. Food and Agriculture Organization of the United Nations.
Gatlin, M. D. , Barrows, T. F., Brown, P., Dabrowski, K., Gaylord, T. G., Hardy, R. W., Herman, E., Hu, G., Krogdahl, A., Nelson, R., Overturf, K., Rust, M. Sealey, W., Skonberg, D., Souze, J. E., Stone, D., Wilson, R. & Wurtele, E. (2007): Expanding the utilization of sustainable plant products in aquafeeds: a review, Aquaculture Research, 38: 551-579.
Hardy, W. R.& Kaushik, S.J.(2021): Fish Nutrition,4.
Hua K , Cobcroft JM, Cole A, et al. The future of aquatic protein: implications for protein sources in aquaculture diets. One Earth. 2019; 1(3), 316-329.
Kaiser, F., Harloff, H.-J., Tressel, R.-P., Kock, T., Schulz, C. (2021): Effects of highly purfied rapeseed protein isolate as fishmeal alternative on nutrient digestibility and growth performance in diets fed to rainbow trout (Oncorhynchus mykiss). Aquaculture Nutrition.
Kasumyan, A. O. & Doving, B. K. (2003): Taste preferences in fishes. Fish and fisheries, 4: 289-347.
Lazzarotto, V., Médale, F., Larroquet, L., & Corraze, G. (2018): Long-term dietary replacement of fishmeal and fish oil in diets for rainbow trout (Oncorhynchus mykiss): Effects on growth, whole body fatty acids and intestinal and hepatic gene expression. PLoS One, 13(1), e0190730.
Marui, T., Evans, R.E., Zielinski, B.& Hara, T.(1983):Gustatory responses of the rainbow trout (Salmo gairdneri) palate to amino acids and derivates. J.Comp. Physiol, 153:423-433.
Nagel, F ., von Danwitz, A., Tusche, K., Kroeckel, S., van Bussel, C. G., Schlachter, M. & Schulz, C. (2012): Nutritional evaluation of rapeseed protein isolates as fish meal substitute for juvenile turbot (Psetta maxima L.)— lmpact on growth performance, body composition, nutrient digestibility, and blood physiology. Aquaculture, 356: 357-364.
Nordrum, S., Krogdahl, A., Rosjo, C., Olli, J. J.& Holm, H.(1999):.Effects of methionine, cysteine and medium chain triglycerides on nutrient digestibility, absorption of amino acids along the intestinal tract and nutrient retention in Atlantic salmon (Salmo salar L.) under pair-feeding regime. Aquaculture,186: 341-360.
Rodehutscord, M., Becker, A., Pack, M. & Pfeffer, E. (1997): Response of Rainbow Trout (Oncorhynchus mykiss) to Supplements of Individual Essential Amino Acids in a Semipurified Diet, Including an Estimate of the Maintenance Requirement for Essential Amino Acids.American Society for Nutritional Sciences.J. Nutr. 126: 1166-1175.
Sarker, P.K. , Kapuscinski AR, Vandenberg GW, Proulx E, Sitek AJ, Thomsen L. (2020): Towards sustainable and ocean-friendly aquafeeds: Evaluating a fish-free feed for rainbow trout (Oncorhynchus mykiss).
Slawski, H. (2012): Rapeseed protein products as fish meal replacement in fish nutrition. Selbstverl. des Inst. für Tierzucht und Tierhaltung.
Yamashita, S., Yamada, T. & Hara, T. J. (2005):Gustatory respones to feeding- and non-feeding-stimulant chemicals, with an emphasis on amino acids, in rainbow trout.Journal of Fish Biology, 68:783-800.