ULTRASOUND-ASSISTED EXTRACTION OF COLLAGEN FROM FISH SKIN: A TIME-EFFICIENT STRATEGY FOR MARINE BY-PRODUCT VALORIZATION
Introduction
The growth of the global population, combined with shifts in dietary patterns, has elevated food security to a matter of critical importance. Within this context, seafood production has experienced significant growth over recent decades, exceeding 180 million tons in 2022 (FAO, 2024). The increase in production has also resulted in a higher volume of by-products, with fish by-products estimated to exceed 20 million tonnes (Yuan et al., 2024). Efforts to reduce the waste of these by-products have led to their use in the production of fishmeal and fish oil . Beyond their use in animal feed production , this biomass contains a significant range of bioactive compounds, offering opportunities to expand the potential applications of these by-products . Collagen falls into this category , with broad applications across food, cosmetic, and medical industries. Collagen serves as a key structural protein in animal tissues and it is particularly abundant in fish by-products, such as skin, scales, and bones . Conventional collagen extraction methods often suffer from inefficiencies, including long pretreatment and extraction times, low extraction yields and the reliance on enzymes (Laasri et al., 2023) . Recent years, environmentally friendly techniques have been developed to significantly enhance extraction efficiency . Among these , ultrasound-assisted extraction (UAE) has emerged as a powerful approach , through cavitation, it can significantly improve collagen extraction times and yields. This study aimed to optimize the ultrasound-assisted extraction of collagen from the skin of Pollachius virens.
Materials and methods
The skin of P. virens was minced and 10 g of tissue were mixed with 150 mL of 0.1 M NaOH to remove non-collagenous proteins. T wo amplitudes (30% and 50%) of sonication were evaluated. Following sonication, the mixtures were incubated at 4 ºC for 24 hours. The soluble protein content in the supernatant was quantified, and the molecular weight distribution of the extracted proteins was analyzed by SDS-PAGE.
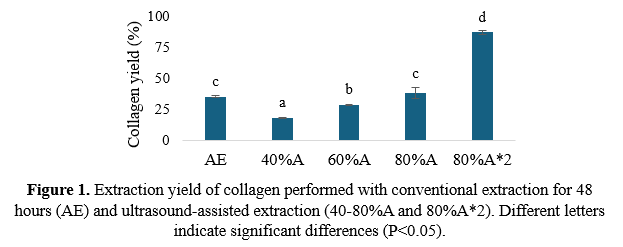
Collagen ultrasound- assisted extraction was then performed on 10 g of pretreated skins with 100 mL of 0.5 M acetic acid at 4 ºC. Both c onventional acid extraction (AE) and ultrasound- assisted extraction at three amplitudes (40%A, 60%A and 80%A) were assessed . Re-extraction of collagen was also evaluated (80%A*2) , the solid residue obtained after the initial extraction at 80% was subjected to a second extraction with 100 mL of fresh acetic acid solution under the same conditions for 10 minutes . The collagen yield was calculated based on hydroxyproline content in the extraction supernatant following the method described by Cissell et al. (2017). The molecular weight distribution of the extracted collagen was analyzed with SDS-PAGE.
Results and discussion
A positive correlation was observed between the amplitude of sonication and the yield of protein extraction during both the pretreatment and collagen extraction stages. During the alkaline pretreatment, sonication at 50% amplitude for 20 minutes achieved a protein yield of 7.7 g per 100 g of dry skin. Notably, extending the treatment by maceration for an additional 24 hours did not further increase the protein concentration, unlike other amplitude and time combinations evaluated.
The molecular weight profile of the proteins extracted indicated that collagen was not co-extracted during this step, suggesting that it remained intact. This approach s ignificantly accelerates the removal of non-collagenous proteins, reducing the required processing time from 6–24 hours to merely 20 minutes without compromising the structural integrity of the collagen present in the skin.
For the subsequent collagen extraction, the effects of varying amplitude were systematically assessed (Figure 1) .
Collagen extraction using ultrasound at 80% amplitude for 20 minutes resulted in a collagen recovery of 38.3%, comparable to the 35.4% obtained through conventional method after 48 hours of extraction . Re-extraction of the solid residue from the initial extraction further increased total final yield to 87.3%, the highest among all tested conditions. S imilar results were obtained for baltic cod ( Gadus morhua) skin after 24 hours of extraction (Sadowska et al., 2003). The molecular weight distribution of the extracted collagen revealed the presence of protein bands corresponding to the α, β, and γ chains characteristic of type I collagen (Shoulders and Raines, 2009).
This methodology offers a efficient route for the valorization of by-products from the aquaculture and fisheries industries, enabling collagen recovery through an extraction process that can be completed in approximately one hour.
References
Cissell, D. D., Link, J. M., Hu, J. C., & Athanasiou, K. A. (2017). A Modified Hydroxyproline Assay Based on Hydrochloric Acid in Ehrlich’s Solution Accurately Measures Tissue Collagen Content.
Food and Agriculture Organization (FAO) of the United Nations (2024). The state of world fisheries and aquaculture.
Laasri , I., Bakkali, M., Torrent, L. M., & Laglaoui , A. (2023). Marine collagen: Unveiling the blue resource-extraction techniques and multifaceted applications. International Journal of Biological Macromolecules, 253, 127253.
Sadowska, M., Kołodziejska , I., & Niecikowska , C. (2003). Isolation of collagen from the skins of Baltic cod (Gadus morhua). Food Chemistry, 81(2), 257–262.
Shoulders, M. D., & Raines, R. T. (2009). Collagen structure and stability. Annual Review of Biochemistry, 78, 929–958.
Yuan, Z., Ye, X., Hou, Z., & Chen, S. (2024). Sustainable utilization of proteins from fish processing by-products: Extraction, biological activities and applications. Trends in Food Science & Technology, 143, 104276.