EVALUATING THE DIGESTIBILITY OF BREWER'S SPENT GRAIN PROTEIN ISOLATE FOR THE RAINBOW TROUT Oncorhynchus mykiss DIET
Introduction
Aquafeed, as major input in aquaculture still relies upon conventional plant protein source like soy. However, the use of soy in aquafeeds are associated with ecological concerns, price volatility, and reduces its availability for direct human consumption (Wang et al., 2023). Brewers spent grain (BSG) is a major agro-industrial byproduct and a potential alternative protein source for soy with the annual production of 40 million tonnes. However, BSG utilization in aquafeed in its raw form is limited due to very low digestibility associated with high fibre content (Karlsen & Skov, 2022) . These limitations can be overcome by extracting BSG protein isolate (PI), leaving behind fibre and other anti-nutritional compounds. Evaluating the digestibility of extracted BSG PI is essential to ensure its nutritional viability. In this study , both in vitro and in vivo digestibility of BSG PI are determined for rainbow trout in comparison with soybean meal to validate the BSG PI as a potential alternative protein source in rainbow diet.
Materials and methods
BSG and protein extraction
Raw BSG was procured from San Miguel Mahou Brewery Ltd., Lleida. Protein extraction was carried out in pilot food industry of IRTA, Monells. Initially, wet BSG biomass was mixed with water at 1:4 (w/w) and heated to 50 0 C. Protein extraction was carried out at pH 11.5 using 2M NaOH 2 hours with constant stirring. The slurry was decanted to separate the liquid phase and reduced the pH to iso-electric point (3.8) The precipitated protein was separated using industrial centrifuge and washed with water to remove excess solutes and neutralized to pH 7 before freeze drying.
In vitro and in vivo Digestibility
For in vitro assay, the stomach and intestine sections of rainbow trout (350 g BW) after 6 hours post-ingestion were used to prepare functional extracts . The acid and alkaline phases of protein hydrolysis was simulated in a two-chambered bioreactor separated by a semi-permeable dialysis membrane with a molecular weight cut of 3,500 Da as described by Lewis et al. (2019). Measurement of acid and alkaline protease by tyrosine release from hydrolysis of haemoglobin (pH 2.5) (Anson 1938) and casein (pH 8.5) (Walter, 1984) respectively. T he duration of the acid and alkaline phases of digestion were 1.5 and 4.5 hours respectively. Samples of BSG protein isolates (58.7% CP) and soy protein concentrate, SPC (61.5% CP) were used for comparison. All assays were performed in quadruplicate. In vivo assay is conducted as part of ongoing replacement trial on rainbow trout. Five diets were prepared by replacing gradient levels of SPC using BSG PI with the addition of inert marker. The in vivo digestibility will be analysed by measuring the ratio of nutrients and marker in the diet and faecal matter.
Results and discussion
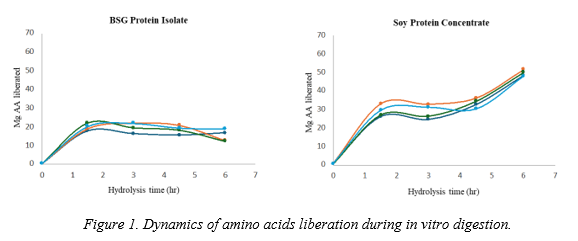
The dynamics of amino acid release during protein hydrolysis of BSG PI and SPC is detailed in Figure 1.
Figure 1 shows hydrolysis profiles of SPC and BSG PI that are generally characterized by a strong initial effect of acid hydrolysis, followed by a stable phase at the beginning of the alkaline phase and a second increase in the release of amino acids after 3 h of alkaline digestion. However, t he hydrolysis profile of the BSG PI was clearly different in both acid alkaline digestion phases. I t showed slightly lower values than SPC during the acid digestion phase. But the drastic effect of hydrolysis was observed at alkaline phase without any increase in amino acid liberation after 3 hours of alkaline digestion.
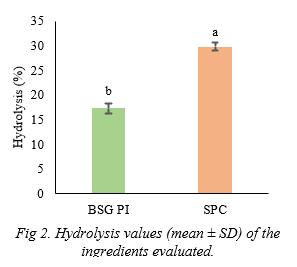
The final hydrolysis values of BSG PI and SPC after in vitro digestion with rainbow trout digestive enzyme extract were presented in Figure 2. Overall, final hydrolysis value of BSG PI (17.3% ± 1.05) was significantly lower (P<0.05) compared to SPC (29.78% ± 0.84) . It is difficult to directly predict the actual in vivo digestibility of the BSG PI via in vitro hydrolysis . Because v arious factors play role in a digestibility in the live organism such as interactions with feed matrix, actual digestion duration, effect of intestinal microbiota etc (Moyano et al., 2015). This is especially obvious in ingredients containing complex polysaccharides. Lower in vitro digestibility of BSG PI in the present study will be validat ed in in vivo by an ongoing feeding trial in terms of key performance indicators associated to growth performance, feed efficiency.
References
Karlsen, F., & Skov, P. V. (2022). Review – Potentials and limitations of utilising brewer’s spent grain as a protein source in aquaculture feeds. Journal of Cleaner Production , 357, 131986. https://doi.org/10.1016/j.jclepro.2022.131986
Moyano, F. J., Saénz de Rodrigáñez , M. A., Díaz, M., & Tacon, A. G. J. (2015). Application of in vitro digestibility methods in aquaculture: Constraints and perspectives. Reviews in Aquaculture , 7(4), 223–242. https://doi.org/10.1111/raq.12065
Wang, J., Chen, L., Xu, J., Ma, S., Liang, X., Wei, Z., Li, D., & Xue, M. (2023). C1 gas protein: A potential protein substitute for advancing aquaculture sustainability. Reviews in Aquaculture , 15(3), 1179–1197. https://doi.org/10.1111/raq.12707