THE EFFECTS OF EPINEPHRINE AND GABA AS CHEMICAL INDUCERS OF METAMORPHOSIS IN Ruditapes decussatus AND Venerupis corrugata
Introduction
Bivalves have a complex life cycle, encompassing a planktonic free-swimming larvae and a benthonic phase1. Metamorphosis is responsible for the transition from larvae to the juvenile stage and represents a critical step in the bivalve life cycle. This process is one of the most challenging phases for bivalve hatcheries due to high mortality and a non-synchronous larval development within a cohort – not all the individuals are physiologically competent to metamorphose at the same time2. To achieve a greater, more rapid and efficient percentage of metamorphosis and consequently improve the production3,4, hatcheries could use synthetic chemical inducers. Among the compounds with an inductive effect on metamorphosis, the amino acid derivative γ-aminobutyric acid (GABA) and the catecholamine epinephrine (EPI) are considered as the most efficient.
Grooved carpet shell (Ruditapes decussatus) and Pullet carpet shell (Venerupis corrugata) are species with high commercial and ecological importance, widely distributed in coastal and estuarine waters throughout Europe, the Mediterranean and North Africa5–7. In Portugal, while R. decussatus, which is produced and harvested mainly in Ria Formosa lagoon, is crucial for aquaculture revenue (representing 90% of the national production), V. corrugata is emerging as a relevant species to Portuguese bivalve aquaculture. Considering the relevance of these species and, in order to improve metamorphosis, with reduced mortality in bivalve hatcheries, the current work evaluated the effects of GABA and EPI, in terms of optimal concentrations, toxicity and exposure time.
Material and methods
Larvae were exposed to three different concentrations (10−2, 10−4 and 10−6 M) of EPI and GABA and filtered sea water (FSW) was used as control. EPI and GABA stock solutions were initially prepared at a concentration of 10-1 M and 10-3 M; stock solutions were then diluted in FSW to reach the experimental concentrations immediately before experiments.
Metamorphosis trials were conducted in glass beakers with 300 ml of FSW, under continuous aeration. Each beaker contained a sieve (150 µm mesh) with approximately 1000 larvae (3 larvae mL-1). Larvae were exposed to different concentrations for 24, 48 and 72 hours. After the exposure period, larvae were rinsed with FSW and 3 aliquots with a minimum of 30 larvae of each beaker were analysed, to evaluate mortality and metamorphic stages. The developmental stages were characterized by the presence of velum, foot and velum, foot, or foot and branchial arches. A larva was considered to have undergone complete metamorphosis when it had lost its velum, was using its foot to crawl and had developed branchial arches. After evaluation, larvae were placed back into the beakers with clean FSW for an additional period of 5, 4 and 3 days, respectively. Metamorphosis rate and mortality were evaluated after each exposure time and at day 6 after the onset of exposure.
Results and Discussion
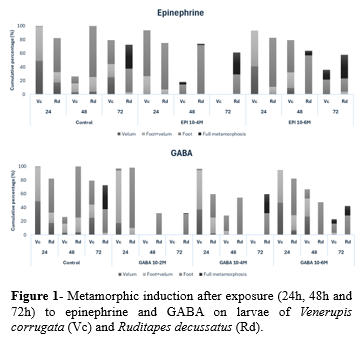
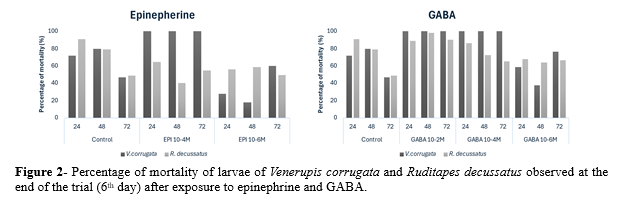
Figure 1 shows the effects of EPI and GABA on the metamorphosis rate of V. corrugata and R. decussatus after each exposure time. In general, high concentrations of both EPI and GABA (10-2 M and 10-4 M) revealed to be toxic to the larvae of both species, regardless of the exposure time (fig. 2). The higher metamorphic rate in V. corrugata and lower mortality were achieved after exposure to EPI 10-6 M (24h and 48h) and GABA 10-6 M (48h) (fig. 1). At 6th day, the 48h-exposure to GABA and EPI 10-6 M induced a higher metamorphosis rate, varying between 62.2% and 82.2%, respectively. Despite its effectiveness in inducing metamorphosis, V. corrugata appeared to be more sensitive to EPI, exhibiting higher mortality rates at certain concentrations.
In contrast, GABA failed to induce metamorphosis in R. decussatus, besides being toxic to the larvae. For this species, 72h-exposure to EPI 10-4 M and 10-6 M induced a higher metamorphic rate, varying between 32.2% and 34.3%, respectively (fig.1). At 6th day of the trial, metamorphosis reached 55.6% and 50.5% in larvae exposed to EPI 10-4 M (48h) and 10-6 M (72h).
These findings highlight the species-specific responses to chemical inducers. Optimal concentration and exposure time vary between species, emphasizing the need to adapt conditions accordingly. Adjusting these parameters may enhance hatchery production by improving metamorphosis efficiency while minimizing larval mortality.
Acknowledgements
This work was financially supported by Portugal’s Recovery and Resilience Plan-European Commission (PRR): Pacto da Bioeconomia Azul-Vertical Bivalves (RE-C05- i01.01)— Agendas/Alianças Mobilizadoras para a Inovação Empresarial.
References
1. Hadfield M, Paul V. 2001. doi:10.1201/9781420036602.ch13
2. O’Connor et al. 2009. doi:10.1111/j.1365-2109.2009.02203.x
3. Doroudi MS, Southgate PC 2002. doi:10.1016/S0044-8486(01)00736-0
4. Mesías-Gansbiller et al. 2008. doi:10.2983/0730-8000(2008)27[261:SBOBSL]2.0.CO;2
5. Cravo et al. 2012. doi:10.1016/j.marenvres.2011.09.012
6. de Sousa et al. 2011. doi:10.1016/j.jembe.2011.06.001
7. Matias et al. 2011. doi:10.1007/s10152-010-0230-3