WET AND DRY MECHANICAL FRACTIONATION OF Tenebrio molitor LARVAE AND THE PROXIMATE COMPOSITION AND IN VITRO DIGESTIBILITY OF THE OBTAINED FRACTIONS
Introduction
Insect s are a promising alternative protein source for aquafeed since they have an adequate nutritional profile, with a high protein content, that can account for more than half of the weight of the dried larvae (Rumpold and Schlüter, 2013) . Insect meals, apart from protein, also contain fat and chitin, which can be separated and further utilized . To obtain these meals, the insect must undergo several processing steps, which may include slaughter, drying, and certain types of fractionations , such as defatting (Ojha et al., 2021) . These processing steps affect different parameters in the obtained meals, such as nutritional, sensory, and hygienic-sanitary quality (Varga et al., 2024) . Mechanical pressing is a relatively cheap, scalable, and economical technique. The fractionation of insects by mechanical dry pressing is frequently applied to eliminate partially or totally the insect fat, while wet pressing is intended to obtain a protein fraction composed mainly of the insect hemocoele and another fraction composed primarily of the insect exoskeleton, rich in chitin. In this study, we evaluated the possible use of each of these fractions as protein-rich meals in aquafeed.
Materials and methods
Tenebrio molitor (TM) larvae were purchased from Artroposfera (Toledo, Spain). Larvae were sacrificed by immersion in liquid nitrogen and stored at -20 °C until further processing. TM larvae were fractionated in a mechanical press at 135 °C. The larvae were freeze-dried (Telstar Lyoquest -55 °C, Barcelona, Spain) prior to the dry-pressing process; a part of the whole dried TM larvae was ground (COM) and stored at -20 °C. DPD fraction was obtained by hot-pressing the dry TM larvae. Wet fractionation was carried out in similar conditions as the dry-pressing process, obtaining a chitinous fraction (WPE) and a fraction consisting mainly of the larval hemocoele (WPH ). WPE and WPH fractions were freeze-dried and stored at -20 °C.
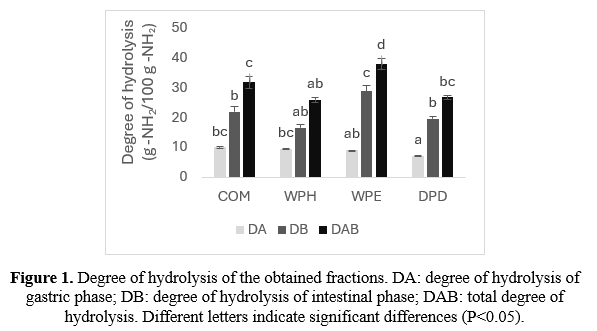
Proximate composition of TM meals was carried out according to the methods described by the Association of Official Analytical Chemists (AOAC, 2005 ). The amino acid profile composition was analyzed with a Biochrom 30+ amino acid analyzer (Biochrom LTD., Cambridge, UK). In vitro protein digestion was performed simulating the gastric and intestine steps of the digestive process, following the modified method described by Varga et al. (2024). T he protein digestibility of TM meal was evaluated using the enzyme extracts from the stomach and intestine of Sparus aurata, and the degree of hydrolysis of gastric phase (DA) and intestinal phase (DB) was expressed as g of -NH2 released with respect to the total -NH 2 in the meal .
Results and discussion
The dry- pressing process yielded 245.2 g of DPD meal and 22.7 g of fat from 1 kg of fresh TM , reaching 845.8 and 78.30 g of DPD and fat, respectively, when calculating with the dry starting material. T he wet-pressing process resulted in 124.5 g of WPE (45 % of dry matter) and 150.4 g (55 % of dry matter) of WPH for each kg of fresh TM used. As for the proximal composition, the protein content calculated from the nitrogen content obtained by Kjeldahl analysis results in a significant increase in crude protein for the WPE and DPD meals compared to the unfractionated insect (COM). The crude fat content was significantly decreased for the WPE and DPD meals with respect to COM, while the WPH fraction accumulated most of the fat resulting from wet pressing. Due to the high content of non-protein nitrogen in insect meals, the Kjeldahl analysis and the choice of the nitrogen conversion factor can lead to errors in the calculation of the protein content. The total amino acid content calculated by HPLC showed that the protein content in WPE accounted for more than two-thirds of the dry weight of the meal.
As shown in Figure 1, the hydrolysis degree results showed that the gastric digestion (DA) resulted in similar values except for DPD, which had a significantly reduced degree of hydrolysis. Furthermore, in the intestinal phase (DB) , the degree of hydrolysis was clearly seen to be significantly increased and reaches its maximum value for WPE with respect to the rest of the treatments. However, when it comes to the total in vitro protein digestibility, this effect lessens.
Results showed that the obtained fractions have high protein levels , satisfactory amino acid profiles, and good in vitro amino acid digestibility. Thus, either the complete insect may be directly included in the diet, or DPD may be applied when the separation of fat is crucial. As for WPH, Kang et al. (2023) have previously suggested its use for human consumption as it had excellent properties as 3D printable food. In the same study , WPE was discarded, which, based on the results of the present work , may be destined to aquafeed as it is a more promising protein source than it was previously believed. T he remaining chitin content can be either separated for further use; or can remain part of the meal, as several authors confirm its beneficial effects in aquafeed .
References
Kang, T. W., Choi, R. Y., Kim, I. W., Lee, E. J., Kim, H. W., & Park, H. J. (2023). Evaluation of feasibility of Tenebrio molitor larval fractions as a meat analog using 3D printing. Innovative Food Science & Emerging Technologies, 89, 103446.
Ojha, S., Bußler, S., Psarianos, M., Rossi, G., Schlüter, O.K., 2021. Edible insect processing pathways and implementation of emerging technologies. Journal of Insects as Food and Feed 7(5), 877-900.
Rumpold, B. A., Schlüter, O. K. (2013). Nutritional composition and safety aspects of edible insects. Molecular nutrition & food research, 57(5), 802-823.
Varga, A. T., Fabrikov, D., Garcı́a, M. V., Jiménez, A. P., Palomares, E. R., Trenzado, C. E., Martín, D., Hernández Llorente, M. D. , Sánchez-Muros, M. J. (2024). How different successive elaboration methods affect Hermetia illucens meals? Macronutrients, in vitro protein digestibility, oxidative status and hygienic-sanitary quality. Journal of Insects as Food and Feed, 1(aop), 1-15.